In sharks, hearing and vibration detection (The Acoustico-Lateralis System) are fundamentally linked. For sharks the inner ears are nestled inside the posterior part of the braincase on top of the head. The only external manifestation of a shark's ears are two small openings on top of the head, just behind the eyes, known as endolymphatic pores.
A shark's main vibration
sensing mechanism is the lateral line, which is visible externally
by a row of tiny pores along each flank. Anteriorly, this system of
pores branches out over the shark's head in complex patterns nested
between and around the electrosensory pores. Despite their apparent
differences, the shark inner ear and lateral line system are based
on the same basic mechanism.
Structure of the Lateral Line and
the Inner Ear
 The functional unit of both the shark inner ear and lateral line is
the hair cell. Each hair cell consists of
a more-or-less globular basal body from one end of which project a
series of cilia (hair-like structures). One of these cilia, called a
klinocilium, is much longer than the
others. The klinocilium extends into a gelatinous dome called a
cupola, which is partially exposed to the external environment. On
the opposite pole of the basal body is a bundle of five or so
sensory nerves. Since water conducts vibrations quite efficiently,
any oscillation in the surrounding liquid medium causes the cupola
to move correspondingly. This movement causes the klinocilium to
bend, which, in turn, provokes the lesser cilia surrounding it to
bend in response (reminiscent of cascading dominos). Bending of a
hair cell's cilia induces an electrical change in the basal body,
which is transmitted - via chemical messengers called
neurotransmitters - to the sensory nerve and on to the brain, where
the stimulus is interpreted as sensation. All these complex
movements and chemical choreography is highly sensitive to even the
tiniest vibration in the surrounding water.
The functional unit of both the shark inner ear and lateral line is
the hair cell. Each hair cell consists of
a more-or-less globular basal body from one end of which project a
series of cilia (hair-like structures). One of these cilia, called a
klinocilium, is much longer than the
others. The klinocilium extends into a gelatinous dome called a
cupola, which is partially exposed to the external environment. On
the opposite pole of the basal body is a bundle of five or so
sensory nerves. Since water conducts vibrations quite efficiently,
any oscillation in the surrounding liquid medium causes the cupola
to move correspondingly. This movement causes the klinocilium to
bend, which, in turn, provokes the lesser cilia surrounding it to
bend in response (reminiscent of cascading dominos). Bending of a
hair cell's cilia induces an electrical change in the basal body,
which is transmitted - via chemical messengers called
neurotransmitters - to the sensory nerve and on to the brain, where
the stimulus is interpreted as sensation. All these complex
movements and chemical choreography is highly sensitive to even the
tiniest vibration in the surrounding water.
The Inner Ear
The shark inner ear is a fluid-filled structure consisting of a
cartilaginous sac to which is attached three semicircular
cartilaginous tubes. These fluid-filled tubes are set at right
angles to one another and are lined with hair cells. Each
semicircular tube responds only to accelerations within the plane
parallel to its orientation. Thus, collectively, the three
semicircular tubes are sensitive to accelerations in all three
geometric planes and grant the shark a simultaneous sense of its
movements in all three-dimensions of its liquid environment.
However, these tubes are not considered to be involved in
sound perception (Carrier et al., 2004).
The saccule, lagena, and utricle are three sensory areas that are
thought to be involved in both balance and sound perception.
They consist of a path of sensory hair cells on an epithelium
overlain by an otconial mass.
The otoconia (Otolith), made of calcium carbonate granules
embedded in a mucopolysaccharide matrix, act as an inertial mass
(Tester et al., 1972).
As in other fishes, the otolith organ is thought to be responsive to
accelerations produced by a sound field, which accelerate the shark
and the sensory macula relative to the otoconial mass (Carrier et
al., 2004). These
otoliths therefore respond to gravity, providing the shark with
information about its orientation in the water, be it head up, head
down, on its side, right-side-up or upside-down.
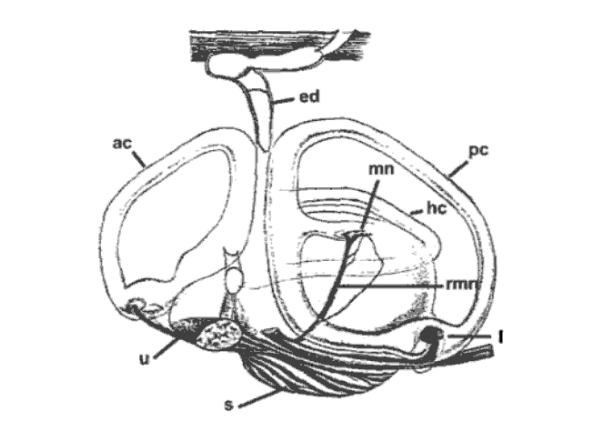
Fig 1: Inner ear of the thornback ray, Raja clavata. ed:
endolymphatic duct; ac: anterior semicircular canal; pc: posterior
semicircular canal; hc: horizontal semicircular canal; s: saccule;
u: utricle; l: lagena; mn: macula neglecta; rmn: amus of VIIIth
nerve innervating macula neglecta (Carrier et al. 2004).
Macula Neglecta
In a fascinating 1981 paper, otolaryngyologist Jeffrey Corwin
reported that in some sharks one of these otolith-equipped parts of
the inner ear, known as the macula neglecta
(because it had long been ignored by sensory physiologists),
responds particularly strongly to vibrations through the top of the
skull. Based on his functional morphology studies of many shark
species, he proposed that the macula neglecta may provide
actively predatory sharks with an enhanced ability to hear sounds
originating from above and in front.
If true, this would grant sharks directional hearing, despite
the close-set arrangement of their inner ear mechanisms. The whole
inner ear structure is connected to the outside of the shark's body
by yet another fluid filled cartilaginous tube. Thus the shark inner
ear is unique among vertebrates in that the fluid inside this organ
is in direct contact with the watery medium outside the animal's
body.
Central Pathways into the CNS (Central
Nervous System)
As in other vertebrates, the ear of the shark is innervated by the
VIIIth cranial (octaval) nerve.
Studies of afferent connections and the physiology of the
octaval nerve form individual end organs (saccule, lagena, utricle
and the macula neglecta) show projections ipsilaterally to five
primary octaval nuclei: magnocellular, descending, posterior,
anterior, and periventricular (Corwin and Northcutt, 1982; Barry,
1987). Much works
remains to be done regarding both the anatomy and neurophysiology of
the CNS.
Sound Waves in Water
Sound
is a multi-stage event that requires four components to occur: a
source of vibration, a transmitting medium, a receiving detector,
and an interpreting nervous system. Sound energy is carried by the
oscillation of particles composing a transmitting medium. In the
case of sharks, the transmitting medium is the water through which
they swim. Thus, distinguishing what a shark hears with its inner
ears from what it senses as vibrations via the lateral line is a
kind of Gordian knot comparable to separating singer and song. As a
result, many shark sensory biologists refer to the combination of
inner ears and lateral lines as the acoustico-lateralis
system. Experiments with various species by Arthur Myrberg,
Donald Nelson, and their co-workers have revealed that sharks are
most attracted to irregular, pulsed sounds of relatively low
frequencies. Field and laboratory experiments have demonstrated that
sharks can hear sounds with frequencies ranging from about 10 Hertz
(cycles per second) to about 800 Hertz, but are most responsive to
sounds less than 375 Hertz.
In contrast, most adult humans can hear sounds ranging from
about 25 Hertz to roughly 16,000 Hertz (young children can hear
sounds up to 25,000 Hertz, but much high-frequency sensitivity is
lost by late adolescence.) Although sharks and humans detect some
low frequency sounds in common, sharks can hear sounds that are
inaudible to us. A shark's hearing is adapted to detecting very
low-frequency vibrations such as those made by a struggling fish.
Sound Detection
 Recently de-classified U.S. Navy studies have revealed that the
ocean is criss-crossed by meandering ribbons of very cold, dense
water surrounded by warmer, less dense water. Since sound travels
more efficiently in dense materials, these liquid ribbons act as
'sound tunnels'. Sound inside these tunnels bounces along like light
in a fiberoptic cable, with very little loss of energy to outside
water masses. During the height of the Cold War, the Navy used a $16
billion system of underwater microphones placed within these
networks of sound tunnels to keep tabs on the positions and
activities of enemy submarines (the system is known by the acronym
SOSUS, for SOund SUrvaillance System).
Some cetologists believe that whales may use these sound tunnels to
communicate across entire ocean basins. Due to its physiological
heat-retaining mechanisms, the White Shark may be able to penetrate
these sound tunnels, listening for the low-frequency sounds of
potential prey inside the cold, dense ribbons of seawater.
Recently de-classified U.S. Navy studies have revealed that the
ocean is criss-crossed by meandering ribbons of very cold, dense
water surrounded by warmer, less dense water. Since sound travels
more efficiently in dense materials, these liquid ribbons act as
'sound tunnels'. Sound inside these tunnels bounces along like light
in a fiberoptic cable, with very little loss of energy to outside
water masses. During the height of the Cold War, the Navy used a $16
billion system of underwater microphones placed within these
networks of sound tunnels to keep tabs on the positions and
activities of enemy submarines (the system is known by the acronym
SOSUS, for SOund SUrvaillance System).
Some cetologists believe that whales may use these sound tunnels to
communicate across entire ocean basins. Due to its physiological
heat-retaining mechanisms, the White Shark may be able to penetrate
these sound tunnels, listening for the low-frequency sounds of
potential prey inside the cold, dense ribbons of seawater.
The Lateral Line (Mechanosense)
The ability to detect movement at multiple scales is essential in
the lives of fishes.
The detection of large tidal currents provides information important
for orientation and navigation, and small-scale flows can reveal the
location of prey, predators, and conspecifics during social
behaviours. The
mechanosensory lateral line system is stimulated by differential
movement between the body and surrounding water, and is used by
fishes to detect both dipole sources (eg: prey) and uniform fields (eg:
currents). This sensory
system functions to mediate behaviours such as rheotaxis
(orientation to water currents), predator avoidance, hydrodynamic
imaging to localise objects, prey detection, and social
communication including schooling and mating (Combs and Montgomery,
1999). In contrast to
the amount of information available on lateral line morphology and
function in bony fishes, relatively little is known about
mechanosensory systems in elasmobranchs.
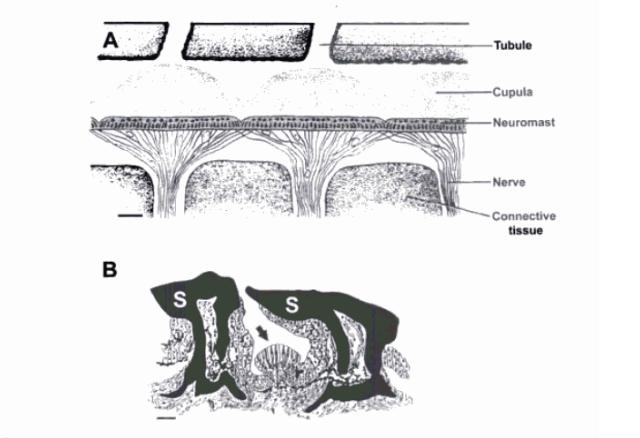
Fig 2: Morphology of the lateral line canal system and superficial
neuromasts in elasmobranchs. (A) Diagramatic longitudinal section of
a pored canal from a juvenile grey reef shark, Carcharhinus
amblyrhynchos. Innervated canal neuromasts are arranged in a
nearly continuous sensory epithelium and covered by gelatinous
cupulae. Pored canals are connected to the environement via
tubules that terminate in openings on the skin surface. Scale bar
150µm. (B) Schematic transverse section of a single
superficial neuromast (pit organ). The sensory neuromast arrow
is positioned between modified scales (S). Scale bar 50 µm. Cupulae
is not shown. (Carrier et al., 2004).
Lateral Line Structure and
Function
The shark lateral line consists of a
fluid-filled, hair cell-lined tube extending along each flank, just
beneath the skin. This tube connects to the external environment via
secondary fluid-filled tubules that branch off from the main tube
and penetrate the skin at regular intervals.
The lateral line system is visible on the surface of the skin
by the presence of small pores known as mechanosensory neuromasts.
Vibrations in the ocean are transmitted by successive fluid
compressions and rarefactions from the secondary tubules to the main
tube. These vibrations then move the gelatinous domes of hair cells
lining the main tube and alert the shark. As the lateral line system
extends along most of a shark's body, it grants the animal a highly
directional sense of movements of potential predators and prey in
its immediate vicinity. The variety in morphological structure and
spatial distribution of the lateral line pores determine functional
parameters such as response properties, distance range of the
system, receptive field area and which component of water motion
(velocity or acceleration) is encoded (Denton and Grey, 1983, 1988).
Sharks that have been temporarily blinded in experiments have
been able to avoid colliding with the wall of the tank which
contained them, apparently by sensing water waves reflected from the
tank wall. Thus, even in highly turbid water, where vision is
all-but useless, a shark can tell exactly where obstacles and other
creatures are, even if it cannot see them.
Lateral Line use in Feeding
Behaviour
 The best known behavioural use of the lateral line in sharks is in
prey detection. Other
uses of the lateral line, particularly in bony fish, include
schooling behaviour, social communication, hydrodynamic imaging,
predator avoidance and rheotaxis.
The concentration of mechanorecpetors on the cephalic region
of sharks and ventral surface of batoids, as well as the low
frequency, close range of the system, indicates an important role in
the detection, localisation and capture of prey.
Swimming and feeding movements of invertebrates and vortex
trails behind swimming fish can produce water movements within the
frequency and sensitivity range of the lateral line system
(Montgomery et al., 1995).
The best known behavioural use of the lateral line in sharks is in
prey detection. Other
uses of the lateral line, particularly in bony fish, include
schooling behaviour, social communication, hydrodynamic imaging,
predator avoidance and rheotaxis.
The concentration of mechanorecpetors on the cephalic region
of sharks and ventral surface of batoids, as well as the low
frequency, close range of the system, indicates an important role in
the detection, localisation and capture of prey.
Swimming and feeding movements of invertebrates and vortex
trails behind swimming fish can produce water movements within the
frequency and sensitivity range of the lateral line system
(Montgomery et al., 1995).
